22 Nov 2022
“Bacteria want to survive and so do humans - it is a permanent race between the two to protect themselves from the other.”
~ Dr Qian Zhang, an antibiotic researcher from Xi’an Jiaotong-Liverpool University ~
You just nipped your finger while chopping an onion. A small cut – you think nothing of it, right? Perhaps you put a plaster on it.
The day after, the cut shows the tell-tale signs of infection – swollen, a bit of pus. No worries, just pop to the doctor for some antibiotics, and it’ll clear up in a few days. You’ll have forgotten about it by next week.
But what if the drugs didn't work? What if the bacteria causing the infection are resistant to the antibiotics and overpower your body’s natural defences? What then?
The number of deaths attributed to antibiotic-resistant bacteria is increasing every year, and the World Health Organization lists it as "one of the biggest threats to global health". Unfortunately, the development of antibiotics is lagging behind the rate of resistance and struggling to catch up. Most antibiotics we use today were developed more than half a century ago.
Why has antibiotic production stalled, and what is the future of antibiotic development? What is causing the increasing number of antibiotic resistance-related deaths? Are we doomed to a post-antibiotic era where even common infections or minor wounds are life-threatening?
Here, we attempt to answer these questions and explain why we are going head-to-head with bacteria in the race to survive.
What are antibiotics, and how do they work?
Antibiotics are chemical substances that can treat infection by inhibiting and killing bacteria.
The antibiotics currently in use work by multiple mechanisms. Some destroy the outer protective layer of bacteria leading to their death; others affect how bacteria work by blocking vital processes they rely on to survive.
Some bacteria – gram-negative bacteria – have an outer layer that makes it very difficult for antibiotics to get inside the cell (picture below). Some drugs, therefore, cannot interfere with the way the bacteria work, making many antibiotics ineffective. Gram-negative bacteria are responsible for most of the deaths caused by resistance to antibiotics.
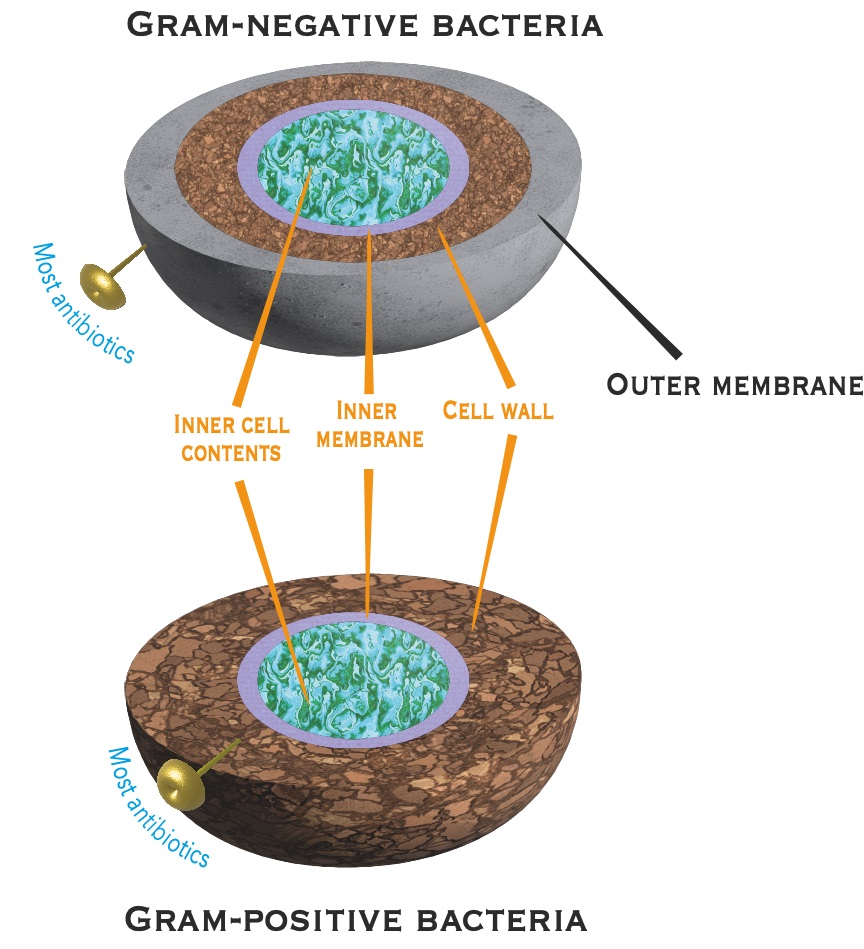
The outer membrane of gram-negative bacteria prevents the majority of antibiotics currently in use from being effective against this type of bacteria. No new mechanisms to enable antibiotics to bypass the outer membrane have been identified for over 20 years.
Sometimes, two or more antibiotics can be given to treat an infection in combination therapy to deal with stubborn bacteria. This is common in the treatment of sepsis and tuberculosis.
Combination therapy may reduce the likelihood of resistance developing, as even if bacteria develop resistance to one antibiotic, the other drug will still kill the bacteria. However, this treatment has considerable disadvantages. The major problem with combination therapy is that both drugs are required to reach their respective targets simultaneously and at optimal concentrations – very difficult to achieve as each person's body will absorb and distribute the drug differently. This gives the bacteria the opportunity to circumvent the treatment.
What is antibiotic resistance, and what are superbugs?
Bacteria have a rapid doubling time and high mutation rate, so when an infection is treated with antibiotics, a mutation may occur in the bacteria that allows it to survive. This mutation is likely to be passed on and reproduced in surviving cells – causing a population to be resistant. Bacteria that become resistant to multiple drugs are dubbed “superbugs”.
Dr Qian Zhang, who is working in antibiotic development at Xi’an Jiaotong-Liverpool University's, says: “Whenever bacteria are introduced to an antibiotic, it is only a matter of time before they develop resistance. Thankfully, most bacteria will take years to develop resistance. However, some develop resistance in just a couple of months or even during the infection of a single person.”
Why is resistance rising?
Resistance to antibiotics is caused by many factors, including poor access to affordable, quality medicines; lack of awareness and knowledge of antibiotic resistance; ineffectual enforcement of legislation; and overuse and misuse of antibiotics in treating both humans and livestock.
There is also a need for more quick diagnostic tests that give detailed information about an infection. At present, when we go to the doctor, it is easy to confirm bacterial infection with a quick blood test. However, the challenge lies in identifying which bacteria is the source of infection and which organ is infected. A lack of detailed information results in doctors prescribing antibiotics that are not targeted in their approach. This, in turn, increases the chance for mutations within bacteria that encode resistance to those antibiotics.
A further problem in the race against antibiotic resistance is that the majority of antibiotics we use today were developed between the 1930s and 1960s. Since 2000, only six new classes of antibiotics have been found, and none act against gram-negative bacteria. This is largely due to the lack of funding for antibiotic development.
Dr Zhang, who previously worked for a pharmaceutical start-up company, says: “Financial markets have a great impact on drug development. Obtaining financial support for the research and development of new drugs has become more and more challenging, especially in the later stage of drug development, such as clinical trials and marketing. These need the financial support of governments and large companies.”
Should we be worried?
Antimicrobial resistance (AMR), which includes antibiotic resistance, is a leading cause of death worldwide, according to a recent study published in the Lancet. The study reported that 3,500 people a day in 2019 died as a result of bacterial resistance to antibiotics - more than malaria or HIV/Aids. This number is only expected to increase. According to a report commissioned by the UK government, AMR could kill up to 10 million people a year by 2050. In response, the World Health Organization has listed AMR as one of the top 10 global threats to humanity.
So is there no hope? What does the future hold?
With the threat of increasing antibiotic resistance and the lack of new antibiotics, combining all available strategies to reduce resistance is needed.
Researchers are working hard to produce new classes of antibiotics that target different components of bacteria and their survival. A promising strategy is being developed by Dr Zhang and Dr Sekar Raju at XJTLU: hybrid antibiotics. These consist of two existing antibiotics that have been chemically linked to create a new single drug, which will help to combat antibiotic resistance and be effective against gram-negative bacteria.
Investment in antibiotic research and development is required for the acquisition of more knowledge of the mechanisms employed by bacteria and antibiotics. Artificial Intelligence will play a significant role in this, as machine-based learning algorithms are already being used to assist in identifying new drugs. Additionally, governments need to strictly regulate the production, sale, and use of antibiotics. Finally, increasing awareness of the dangers and causes of antibiotic resistance is paramount to reducing this global threat and not falling behind in the race against antibiotic resistance.
What can you do to reduce the risk of antibiotic resistance?
- Don’t abuse antibiotics. Ask a doctor for advice before taking them. Doctors can check if you have a bacterial infection.
- Avoid sharing your antibiotic medication.
- Take the whole course of antibiotics. Don’t stop early!
For more information visit the WHO, FAO and UN antibiotic web pages.
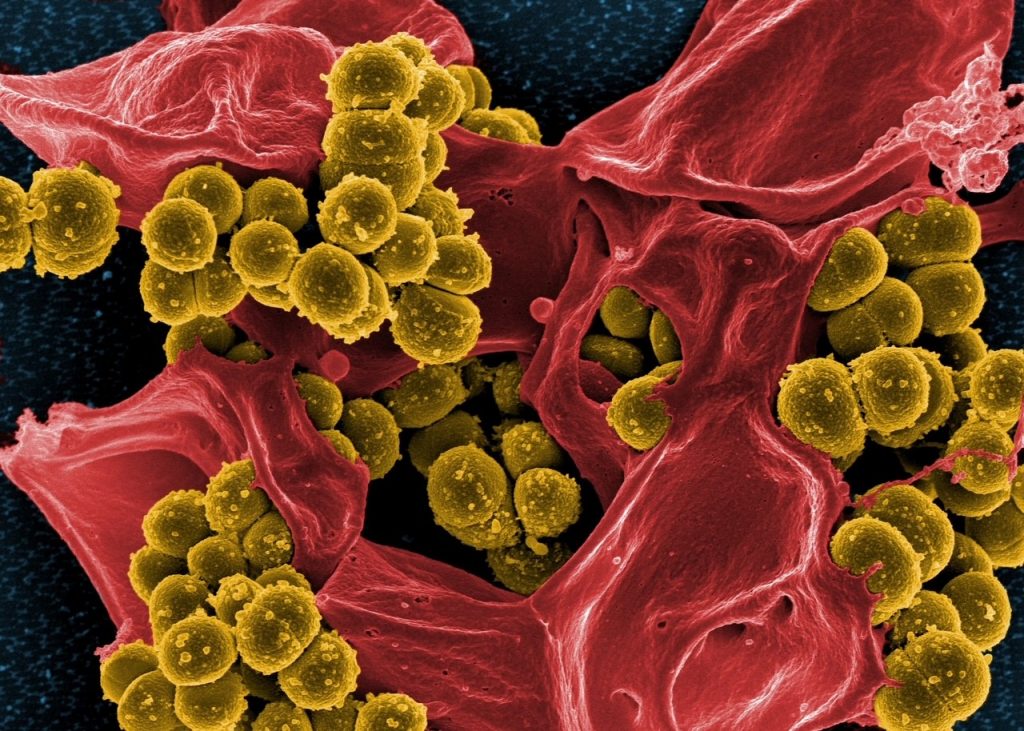
Scanning electron micrograph of methicillin-resistant Staphylococcus aureus bacteria (yellow) and a dead human white blood cell (coloured red).
By Catherine Diamond and Luyao Wang
Edited by Patricia Pieterse
22 Nov 2022
RELATED NEWS
Researchers use bacteria to enhance concrete resistance
Concrete, with its low cost, good in compressive strength, and manufacturing convenience, is one of the most widely used materials in construction. However, ...
Learn more

Eating sea squirts may reverse the signs of ageing, study shows
If you have ever looked in the mirror and seen greying hair and wrinkles or forgotten the name of a close friend, you’d be forgiven for wishing for a pill th...
Learn more

XJTLU researchers use new material to improve solar cells
The current global energy crisis has emphasised the urgency to reduce our reliance on non-renewable power. Solar power is one of the best strategies for gene...
Learn more








