07 Apr 2026
Recently, Professor Li Yang and her team from Xi’an Jiaotong-Liverpool University, together with researchers from the University of Liverpool, published a study offering a new approach to improving the safety, lifespan and stability of lithium-metal batteries.
The research, published in the journal Advanced Functional Materials, proposes a new strategy to regulate lithium ion transportation using porosity and acidity in molecular sieves.
According to Professor Li Yang, one of the paper’s corresponding authors, lithium metal is widely seen as one of the most promising anode materials for next-generation high-energy-density batteries because of its high energy-storage capacity.
However, one major barrier has limited its large-scale use for years: lithium dendrites. These tree-like metal structures can keep growing inside a battery, raising the risk of short circuits, shortening battery life, and causing safety problems. To combat this, battery researchers have focused on improving separator performance, so that separators can better suppress the growth of lithium dendrites while also offering greater mechanical strength.
Professor Yang explains that although a separator is very thin, it plays a key role in a battery. It keeps the cathode and anode apart to prevent short circuits, and also affects how lithium ions move through the battery.
Professor Yang says: “The separator we developed acts as a dedicated fast track for lithium ions while also regulating the lithium-ion pairing structure. It helps them move faster and more steadily inside the battery, while also creating a protective layer to reduce the formation of dangerous lithium dendrites.”
A way to guide lithium ions
The team found that although conventional separators are widely used, they have limitations in high-performance lithium-metal batteries. These include poor thermal stability, limited compatibility with electrolytes, and a lack of active control over lithium-ion transport.
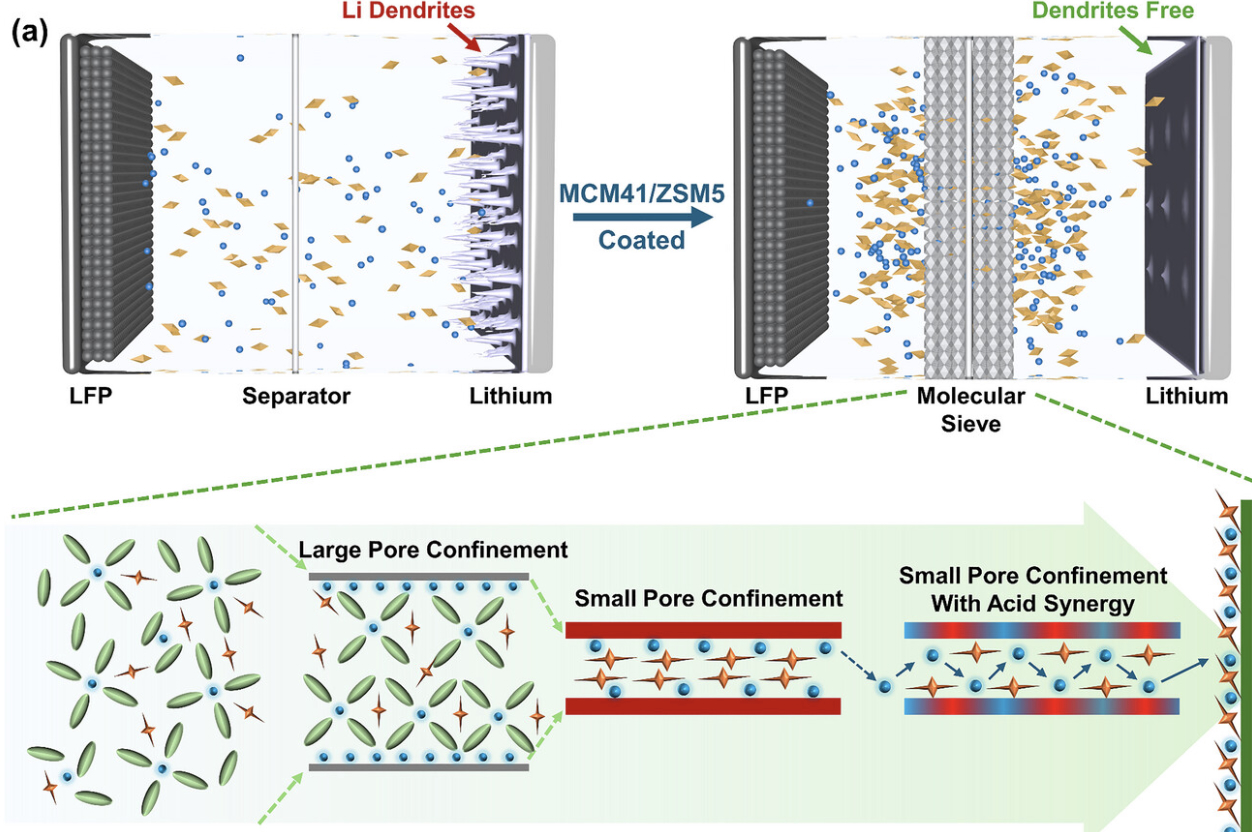
To address this, the researchers turned to molecular sieve materials, which are widely used in chemical engineering. Molecular sieves are often used to adsorb and separate molecules. In this study, the team introduces them into battery separator design to take on a new role in regulating ion transport.
“You can think of a molecular sieve as a security checkpoint,” Professor Yang says. “It acts like an intelligent gate in a passage, allowing ions to move forward in a more orderly way while reducing interference from other components, so the transport process becomes smoother.”
The schematic diagram of the molecular sieve functional membrane controlling the transmission of solvent molecules, lithium ions and anions through its pore size and acidity.
A combined effect
The main innovation of the study is its systematic combination of molecular sieve porosity and acidity to regulate lithium-ion behaviour more precisely. The team built a series of composite separators based on different molecular sieves and compared how different pore sizes and surface properties affect battery performance.
Professor Yang explains: “Porosity works like a sieve. It filters out larger molecules and allows only small ions to pass through. Acidity works like a magnet. It attracts anions and releases more free lithium ions. Only when the two work together can the system both filter accurately and release more ions, creating a combined effect greater than either one alone.”
In simple terms, the small-pore structure helps screen and constrain molecular movement inside the battery, while acidic sites on the material surface help reduce interference and release more lithium ions for efficient transport. When these two effects work together, lithium ions move more smoothly, and a more stable protective layer forms inside the battery, reducing the growth of lithium dendrites.
Experiments show that compared with conventional separators, the new molecular-sieve separator significantly improves cycling stability and battery life. Even under fast charging and discharging and relatively demanding operating conditions, the battery retains about 95.7% of its capacity after 2,900 charge-discharge cycles, while maintaining highly stable performance throughout the process. By contrast, batteries using conventional separators develop serious short circuits within 1,000 cycles.
“This means the battery could operate stably for nearly eight years under a usage pattern of almost one charge per day,” Professor Yang says.
Potential for future applications
In addition to its long cycle life, the study also finds that with the new separator, the surface of the lithium anode becomes smoother and denser, with almost no visible signs of dangerous dendrite growth. At the same time, the protective layer on the electrode surface is more stable, which is one of the key reasons the battery offers greater safety and a longer lifespan.
“This finding may support future applications in areas that require high battery safety and long service life, such as electric vehicles, energy storage stations, portable devices and spacecraft. However, moving from laboratory results to industrial use still requires overcoming practical challenges, including large-scale production, cost control and compatibility with existing battery manufacturing lines,” Professor Yang says.
The team now hopes to develop more separator materials with similar synergistic effects and explore their potential in other battery systems, opening up new possibilities for the development of related energy-storage technologies and offering a new idea for designing safer and longer-lasting lithium-metal batteries.
The paper, titled “Confined Ion Regulation via Synergy of Porosity and Acidity in Molecular Sieves for Lithium-Metal Batteries”, was first-authored by Jingchao Zhang, a PhD student, and Dr Jianbo Li, a co-supervised postdoctoral researcher, from the Advanced Materials Research Centre and the School of Science. The corresponding authors are Professor Li Yang from the School of Science and Dr Chenguang Liu from the School of Robotics. This research was supported by the Advanced Materials Research Centre. The full paper can be read here.
By Luyao Wang
Edited by Patricia Pieterse
07 Apr 2026








